Ecological Darwinism or Preliminary Answers to Some Crucial though Seldom Asked Questions
Almanac: History & Mathematics:Big History Aspects
Abstract
The author asserts that evolutionary regularities might be deduced from principles of life's functioning. First of all, the latter should describe the part-whole relationships and control mechanisms. The author suggests supplementing the concept of struggle for existence with the concept of functional hierarchy: no solitary individual or species is functionally autonomous, life as we know it can exist only in the form of a nutrient cycle. Only two purely biotic forces – ‘biotic attraction’ and ‘biotic repulsion’ – act in the living world. The first one maintains and increases diversity and organizes solitary parts into systems integrated to a greater or lesser degree. The second one, in the form of competition, lessens biodiversity but at the same time provides life with necessary plasticity. On that basis, tentative answers to the following questions are given: (1) Why does life exhibit such a peculiar organization: with strong integration at lower levels of organization and weak integration at the higher ones?; (2) Why did particular species and guilds appear on the evolutionary stage at that particular time and not at any other?; (3) Why was the functional structure of ecosystems prone to convergence despite a multitude of stochastic factors?
Keywords: ecological Darwinism, biology, selection, ecosystems.
Introduction
As a matter of fact, Darwin's theory on natural selection consists of two constituent parts: the ecological and the genetic one. The first of them (‘struggle for existence’) deals with a surplus in offspring and unfavourable environmental factors, which cause mortality of the former. The part of genetics focuses on undirected variability and inheritance of selected traits. Geneticists of the 20th century specified and elaborated the latter part of the theory. Meanwhile, the first part of the theory fell into the hands of ecologists and also underwent elaboration. However, ecologists did not restrict themselves to ‘struggle for existence’ and created something that was new in principle, i.e. the ecosystem conception. Many of its statements are still valuable to date. Strange as it is, until recently evolutionists have hardly made any use of this part of ecology, and it lingered where it was created. It is strange because when referring to any hypothetico-deductive theory (Darwinism is undoubtedly such a theory), it is advisable from time to time to revert to its original postulates to verify if they are in agreement with new data (Popper 1959). The ecological part of the natural selection theory deals with the way organisms react to the environment. If these relations are not restricted just to ‘struggle for existence’, it is not only possible but advisable to supplement the premises of the theory with the new ones. In turn, conclusions following from the original statements will change. So, if we want to have a more profound evolutionary theory which better corresponds to the present-day achievements, we must revert to Darwin's original premises and reassess them not only from the viewpoint of genetics but also from that of ecology.
I am pretty sure that the need for a new evolutionary theory is felt most strongly by those who cannot find answers to the questions concerning the ‘essence’ of life and the main regularities of its functioning and evolution. It is quite possible that the majority of biologists believe that all the questions of fundamental significance have already been answered. I assume such a viewpoint to be excessively superficial. I think that it is only with the help of ecological theory that it is possible to give an answer to many fundamental questions which traditional biology did not even raise. For instance:
· Why does life exhibit such a peculiar organization: with strong integration at lower levels of organization and weak integration at the higher ones?
· Why did particular species and guilds appear on the evolutionary stage at that particular time and not at any other?
· Why was the functional structure of ecosystems prone to convergence despite a multitude of stochastic factors?
The material presented in this survey raises hope that answers, tentative as they are, to these and the like questions may be perceived in the near future. Such ‘ecologizing’ of Darwinism is likely to benefit not only this theory but ecology itself as well.
The Possible Methodological Framework for the Future Evolutionary Biology
Let us start by formulating the main methodological principles, i.e. particular ‘recipes’ which should be followed if we want to guarantee success in devising a more extensive synthesis.
It is usually claimed that populations and ecosystems are complicated and difficult to investigate objects; subjects of study of molecular biology and especially biophysics are less complicated by comparison, therefore progress in these spheres is greater. My opinion is slightly different: the complexity of life phenomena is largely the creation of our own minds and is a consequence of research methods applied that are not entirely adequate. At first glance it seems that life is objectively complex only if we measure complexity in terms of heterogeneity or the variety of structures. Biologists who talk about the complexity of life very often appeal to the abundance of links as well. And in this regard they are right again: multicausality is the result of this abundance. However, it is quite possible that life will lose most of its complexity when the new method of logical simplification that has been ignored heretofore is applied. I will try to propose the guidelines for such simplification.
I would define recipe 1 in the following way: the deductive method, especially thought experiments, should not be avoided while pursuing wider biological synthesis. Biologists have largely ignored the deductive method. Undoubtedly, this has led to adverse consequences and it is hard to explain exactly why this has happened. It is possible that the majority of biologists identify the deductive method with axiomatic methods, which are unacceptable to most biologists, and maybe not without good reason. I am also sure that almost all biologists associate deduction with mathematical methods, which is also a real misunderstanding. We use deduction in our everyday lives, and without it we would be simply unable to understand each other. Although Darwin generated a number of ideas through deduction and without using mathematics, most biologists understand and appreciate their value.
recipe 2 is as follows: while proceeding with the development of evolutionary theories (deriving evolutionary regularities from functional ones) initiated by Darwin, it is necessary to revise not only the conception of the ‘struggle for existence’ but also the attitude towards the nature of intra-organism links adopted in Darwin's lifetime. The main question is: what is the nature of the ‘part-whole’ relationship at every level of organization, starting with macromolecules and ending with ecosystems. Such union of functional biology and evolutionary biology makes it possible to explain the maximum number of phenomena on the basis of the minimum number of statements. This is the main purpose of any logical simplification.
Recipe 3: while analyzing the causes of biological phenomena, it is recommended that the widest implications of cause-effect relationships be given consideration. Of course, this recipe can be useful only to a theorist pursuing synthesis. The well-established tradition, which encourages interest only in direct relations is potentially disastrous to theoretical work aimed at synthesis. However, it must be noted that a physicist or chemist would hardly admire this recipe, and many may claim that it would make the biological view of the world even more complex, but we should not be concerned with that; as it becomes clear through causal analysis of this type, that biotic connections are ‘neatly built’ and characterized by a particular hierarchy. Using this recipe, it has been established, for example, that it is not only plants that participate in the process of photosynthesis (as it is usually considered) but almost all the local ecological community (Lekevičius 1985).
Recipe 4 recommends using a qualitative or conceptual method of modeling. Even though this method is used in biology quite widely, I suppose I should describe it in greater detail. This modeling can be viewed as intermediate between verbal and mathematical modeling. Darwin's theory of natural selection can be considered as a typical verbal model. To transform it into a qualitative model, it is necessary to formulate and define its original terms and statements (premises) strictly. Qualitative models would include graph diagrams indicating only trends and various kinds of diagrams displaying connections between objects and phenomena and the like. The disadvantage of this modeling is its insufficient precision. However, there are particular advantages to using this modeling also: it does not restrict the modeler to any particular mathematical apparatus, the researcher therefore has much more freedom to raise questions and suggest hypotheses than he/she would have if mathematical modeling was employed. This kind of modeling additionally offers the opportunity to ‘cover the uncoverable’ (see Recipe 3). Furthermore, it allows adapting the method to the aims and research objects rather than the other way round, which is often the case, especially in ecology and evolutionary biology when mathematical methods are employed. Mathematical modeling could even follow from qualitative modeling as it usually happens in physics.
According to the instructions of Recipe 5, one of the main ways to engage in logical simplification is to adopt the functional point of view. This rule is based on the fact that the variety of structures (macromolecules, sub-cell organelles, cells, organs, genotypes, phenotypes, and species) is far richer than the variety of roles or functions that these structures perform. From the structuralist point of view, every enzyme is fairly complicated, and in order to describe this variety in detail, an exhaustive and difficult research effort is required. Meanwhile, its function is comparatively easy to identify and can be defined in a single sentence. Additionally, simplicity can be seen in the fact that the organization of life in its entirety is based on a certain hierarchical system: general functions, such as local nutrient cycling, can be fulfilled only through partial functions that are performed by individual guilds and species of the ecosystem. To attain simplicity, it is necessary to abstract from details. In our situation abstraction is easy to achieve because nature seems to have already provided it for us through the manifestation of a few functions (‘emergent’ features) present at the highest levels of organization. This sharply contrasts with the abundance of partial functions found at lower levels.
The Nature of ‘Part-Whole’ Relations
Let us conduct a thought experiment: let us imagine an organ in isolation and try to find an answer to the question as to how long it could survive without the appropriate biotic context, in this case – the organism (1). Let us now do the same with an individual animal or plant (2) and any population (3). The results are going to be more or less as follows. The organ will cease to function very soon, the individual will, however, survive for a longer period of time, and the population will survive still longer. It does not matter if you isolate an individual plant from its biotic environment, the whole population, or all ‘autotrophs’ of a particular ecosystem. The result in all cases is going to be the same – loss of life. The only difference, when compared with animals, might lie in the fact that some ‘autotrophs’, when isolated from detritivores (= decomposers) will be able to survive for a longer time – up to a few years or a few decades, depending on the amount of supply of inorganic nutrients available at the beginning of the experiment.
This fact illustrates that
functional autonomy is not characteristic of any of these structures. If the
biotic environment is eliminated they cannot be consi-
dered to be alive, in a sense. Following similar logic, biologists have claimed
that viruses are not live organisms since they can only reproduce using the nuclear
apparatus of a host's cell. This verdict does not seem to be controversial, but
then, using this same logic, we may ask why we consider a deer or a lion, for
example, to be alive.
Having conducted these experiments you will be forced to admit that in some sense the main feature of being alive, i.e. functionally autonomous, is only typical of an ecosystem, since life cannot last independently without nutrient cycling. The formula ‘only an ecosystem is living’ should be interpreted in the following way: a nutrient cycle is an emergent feature shaped by the coordinated activity of the whole ecological community (at least that of ‘autotrophs’ and detritivores). Let us call the local cycle and the energy flow that follows it the global function. The activities of individual guilds or species, then, could be treated as partial, or minor, functions, those of separate individuals – as even smaller functions, and so on, until we come to the functions of macromolecules. Eventually, we arrive at what systemologists refer to as a functional hierarchy. This concept might be more convenient to use, but it essentially means the same as functional dependence. It follows then that it is not simply integrity that is characteristic of life, but integrity based on mutualism, or links of reciprocal dependence. A biological species is not an aim per se, as it is usually assumed, but also a means.
This can still be expressed in a different way, by adopting the concept of labour division that Darwin (1872) was so very fond of: nutrient cycles are the outcome of common activity of individual species that are involved in labour of a narrower kind. Each of them performs a different operation. Again, specialization in reproductive or any other function is available within the population framework. This is directly analogous to the division of labour typified by the arrangement of organs, cells and macromolecules in a single living being.
There might be, in fact, several varieties of hierarchy. One kind of hierarchy is typical of clockwork mechanisms, for example, another kind of hierarchy – of a multi-cellular organism, and still another one – of a population or a community/ecosystem. Clock-parts have no capacity for reproduction. It might be claimed that the structure of a clock is therefore inflexible and completely inert, and that its parts therefore lack the ‘freedom’ to pursue their own self-interest. A multi-cellular organism has a more or less different hierarchy of functions, with cells of an organism capable of proliferating and therefore having some freedom to pursue their self-interest, although minimally. An organism is flexible and can adapt to the ever changing environment (physiological and biochemical adaptation), the freedom to pursue self-interest at the level of sub-individual structures being a prerequisite for this. Cells might even compete with each other while pursuing their individual ‘goals’; experiments with chimeras largely contributed to this conclusion (McLaren 1976; see also the review by Lekevičius 1986: сh. 3.4). As far as individuals and species are concerned, they possess even greater freedom. This freedom is so great that the majority of ecologists still conceive of nature as being governed by competition (‘biotic repulsion’) and still argue that interdependence (‘biotic attraction’) does not exist at all at the levels of population and ecological community; and even if it does, this interdependence can be ignored. Extensive biological data show that individuals and species use this freedom for their own ‘purposes’ which are usually related to generating even larger populations.
To my mind, the analogy of the two-faced Janus can be used (Koestler 1967) to reveal the essence of the part-whole relationship. The side of his face that is turned upwards, towards the higher level of organization, shines with obedience and devotion, whereas the one turned downwards is the face of an individual who recognizes only his own objectives. Biosystems can be regarded not only as multilevel, but also as multigoal systems (Mesarović et al. 1970) in the sense that the goals of individuals and species do not necessarily have to be the same. Their respective objectives might even be in conflict with one another, which is what we usually observe in nature. On the other hand, the fairly great freedom of action that is noticeable at these levels seems to be very useful to ecosystems when they have to adapt to drastic and unpredictable environmental changes. So, the functional hierarchy in nature is not rigid or stiff. From a functional point of view, biotic components, starting with cells and ending with species, do not only depend on each other, but are also conditionally independent, as they cooperate and compete with each other simultaneously. The interaction of these two opposite forces, ‘biotic attraction’ and ‘biotic repulsion’, determines the behaviour of life and its evolution.
The functional hierarchy cannot be realized without an adequate hierarchy of control. However, there exists no control device at these levels of organization, which is similar to that of multi-cellular organisms. Many people may consider this situation absurd, but this is nothing new for experts in systems theory. This type of control has been termed as diffusive or passive (Novoseltsev 1978; Lekevičius 1986). It is achieved through the interaction of sub-systems, whose behaviour towards control is the same. During these interactions, certain constraints (positive or negative feedbacks), helping the whole to control its constituent parts, emerge. These constraints usually evolve because not all combinations of subsystems or their activities can ensure stability. Populations and ecosystems therefore adjust on their own, without any external contribution. It means that joint efforts help ecological communities not only to support local nutrient cycles, but also to ensure their conditional independence from various kinds of inner and external perturbations. In other words, global parameters, vitally important for the whole biota, are homeorhetic because of self-organization and self-regulation. Nutrient cycles are the most highly buffered features of life (Lekevičius 1986).
It might be even easier to understand how the ecosystem's functioning is controlled by considering an analogous example of capitalist economy, the laissez-faire mode in particular. The forces of ‘repulsion’ and ‘attraction’ in capitalist economy are almost equal in power, their counterbalance being nearly the same as that in nature. The initiative and the right to decide belong to individuals. Although, as a rule, they pursue self-interest rather than the interest of society, the society, guided by an ‘invisible hand’ (in fact – by the market), inevitably tends towards the universal well-being. This conception became popular in England as the paradox of private vices and public benefits.
Incidentally, Darwin was probably the first to notice parallels between the organization of economy and that of nature. They have also recently been discussed by Salthe (1985) and Lekevičius (2009a). Naturally enough, using analogies is not an appropriate way to explain something. However, I do think that it may be beneficial for the clarification of statements.
Why does life exhibit such a peculiar organization: with strong integration at lower levels of organization and weak integration at the higher ones? To answer this question, let us think what animate nature would look like if individuals of the same and different species only cooperated, i.e. if competition as a phenomenon completely disappeared. A preliminary answer to this and some other questions of a similar kind has been provided by GAT, the general adaptation theory (Conrad 1983; Lekevičius 1986, 1997). According to the theory, if this hypothetical situation came into being, we would probably have both ecosystems and nutrient cycles. In fact, these would not be typical ecosystems; they would have a much greater degree of integration – somewhat comparable to today's coral reef ecosystems. These ecosystem-superorganisms would perform their vital functions incredibly effectively, but would fall to pieces like a giant with clay feet as soon as the first unusual environmental change took place. Specialization and integration allow maximizing the degree of adaptation, but that is incompatible with maximum adaptability. The capacity for disintegration and the conditional freedom of subsystems are essential attributes of life on this planet, where environmental conditions are continually changing to a great degree and are very often unpredictable.
What would happen in the opposite situation, i.e. if these relationships were only of a competitive type? I think that the most likely final outcome would be that only one species would exist in any given location at any given time; i.e. the one that would have replaced all the rest species, those that are not so well adapted to struggling for existence. And within this species, a single (‘wild’) genotype that has replaced all the genotypes of lower adaptive value would exist. Naturally, there would be no ecological communities or ecosystems under these circumstances. However, as it has already been mentioned, this sort of life would have no chances of survival since none of the species can maintain a nutrient cycle on its own. To summarize, it might be claimed that life has chosen a compromise between two incompatible strategies – to be maximally efficient and not to compromise adaptability. This compromise must have conditioned the long-term existence of life. However, the problem with this kind of an answer lies in its teleological implications. We could arrive at a far better answer if we discovered the evolutionary processes through which this form of life could emerge.
On Evolutionary Interdependence of Individuals and Species
Having applied the methodological recipe advocated by us (evolutionary regularities can be deduced from principles of functioning – recipe 2), we come to the conclusion that even when evolving, individuals and species cannot have autonomy. Functional dependence inevitably leads to evolutionary dependence. It is clear that the most obvious manifestation of this regularity is likely to be observed in cases of cooperation and mutualism. For instance, it is clear that organs of a multicellular organism can evolve only in a coordinated manner. Otherwise, the integrity and vitality of an organism will be destroyed. Similarly, the evolution of members of the same population, which are bound by relations of interdependence, cannot be uncoordinated. For instance, an uninterelated evolution of males and females of the same population is difficult to imagine. In these cases, loss of coadaptation is equivalent to population extinction. It is also obvious that evolutionary changes in species bounded by mutualistic relationships cannot be uncoordinated either. For example, such coevolution should be characteristic of flowering plants and insects pollinating them. The same holds true for the relations between producers and detritivores of the same ecosystem: both these ecological groups should inevitably affect the evolution of each other, as they are mutually dependent. In short, coevolution or coordinated evolution is the inevitable outcome of functional dependence.
Populations of prey and its predators like those of hosts and their parasites also coevolve. For instance, extensive available evidence shows that prey/host populations accumulate features reducing exploiter-induced mortality. In length of time, the latter, in their own turn, acquire features enabling them to continue the exploitation of their usual prey/host. It is clear that the initiative should not necessarily come from exploited populations. Such coevolution usually leads to moderate exploitation. And only in case of moderate exploitation, we have the right to assert that both partners are coadapted. In this context, I suppose, I do not violate the terminological discipline, as, to my knowledge, the terms ‘coevolution’ and ‘coadaptation’ are treated in this way by the majority of users.
In my opinion, the evolution of most species was and still is restricted from every side, as the ecospace nearest to them was and still is occupied by other species well adapted to their environment. Species do not exist in some kind of ecological vacuum – both their functioning freedom and that of evolution are restricted. Therefore prohibitions have always outnumbered permissions. Stabilizing selection and evolutionary stasis are daily routine of animate nature. Many of the evolutionarily old species can be treated as living fossils, which is not because they lack variability, but because other species (most often those that have emerged later) did not leave free ecospace for the new variations to penetrate. This approach, in my opinion, considerably differs from the opinion that has been dominant for a long time. According to that view, the rate and success of evolution are predetermined only by genetic variability; and maybe also by climatic and edaphic conditions and geographical isolation.
In this context, permission is understood as a vacant niche, and, more exactly, as a vacant environmental niche. Two terms are used in ecology: an ecological or Hutchinsonian niche, on the one hand, and an environmental niche, on the other. The first one is understood as the totality of needs. The ecologists using the term ‘an environmental niche’, first of all, have environmental conditions in mind, which, in their opinion, can exist and exist independently of organisms. It is only they who use the term ‘a vacant niche’. They understand a vacant niche as potentially usable resources. Solar energy having no consumer, some organic or inorganic substance as a potential source of energy, electrons or carbon can be taken as examples of such resources. Of course, a live organism also can be treated as a vacant niche if it does not have consumers (predators or parasites) (for more information about the vacant niche concept see the survey by Lekevičius [2009b]).
An occupied niche can be viewed as prohibition for another species to occupy it. However, this prohibition can be overcome in cases of successful innovations or immigration of stronger competitors. Sometimes it is more expedient to replace this term (prohibition) by the term ‘constraints’, which may sound less categorical. Of course, in addition to constraints associated with the availability or absence of resources, there are other types of constraints, such as thermodynamic, climatic or edaphic constraints. Their evolutionary impact is quite well-known and we are going to discuss them as well. In essence, prohibition can be associated with the fact that not all evolutionary trajectories ensure stability of living systems. For instance, ecosystem-level constraints are, first of all, negative feedbacks, which do not permit species to evolve in such a way that the local nutrient cycle should be disturbed. So, it is possible then to view permissions as positive feedbacks and prohibitions as negative ones.
Evolutionary Assembly of Ecosystems
Ecosystem ‘assembly rules’ may be formulated in the following way (Lekevičius 2002). It is quite possible that since the very moment of life appearance there existed quite a simple mechanism by which ecosystems and nutrient cycles were formed – end products of some organisms' metabolism turned into waste, i.e. into resources potentially usable though used by nobody. Such vacant niches provoked the evolution of organisms capable of exploiting those resources. The final result was that end products of detritivores' metabolism became primary materials for producers. The formation of ecological pyramids should have followed a similar pattern: producers provoked the evolution of herbivores, the latter – that of primary predators, and so on and so forth until eventually the evolution produced common to us pyramids with large predators at the top.
So, vacant niches not only stimulate diversification, but also determine its direction. And this fact, most probably, witnesses causality. This idea can be viewed as a keystone of evolutionary theory because it is not so difficult to explain, and, at least partly, predict results of diversification from data on vacant niches.
In order to clarify the vacant niche concept and its usage, I have constructed a table demonstrating some steps in ecosystem evolution.
The first terrestrial organisms should have probably been heterotrophs. The main shortcoming of the first ecosystem was that decomposition was carried out far more intensively than the chemical synthesis of organic matter. This misbalance might have caused the very first in the history of life ecological crisis, which finished with the rise of the first producers. The latter could have been green and purple non-sulfur bacteria, which carried out anoxygenic photosynthesis. They used organic compounds as a source of hydrogen (electrons).
Along with these bacteria, detritus-decomposing ones, too, are likely to have been involved in local nutrient cycles of that time. Their emergence and diversity was determined by the diversity of organic substances present in detritus. Already at that time cycles must have been non-waste, and decomposition was carried out to the very inorganic nutrients.
Table. Some of the vacant niches/adaptive zones that existed in the Archean and Proterozoic, and their occupants
|
Description of vacant |
Hypothetic occupants |
|
Organic substances as donors of energy, electrons and carbon. Organic substances as final electron acceptors |
Protobionts |
|
Light as an energy donor, H2S/H2O as an electron donor and CO2 as a donor of carbon |
Green and purple sulphur bacteria, cyanobacteria |
|
Detritus as an energy, electron and carbon donor. So and SO42– as final electron acceptors |
Sulphur- and sulphate-reducing bacteria |
|
Fe2+, Mn2+, H2S, CO, H2, CH4, NH4+ as energy and electron donors, CO2 as a carbon donor. O2 as a final electron acceptor |
Aerobic chemolithoautotrophs |
|
Detritus as an energy, electron and carbon donor. NO3– as a final electron acceptor |
Denitrifying bacteria |
|
Detritus as an energy, electron and carbon donor. O2 as a final electron acceptor |
Aerobic decomposers |
|
Biomass as an energy, electron and carbon donor. O2 as a final electron acceptor |
Protists as ‘herbivores’ and decomposer-eaters |
|
Biomass (‘herbivorous’ and decomposer-eating protists) |
Protists as primary predators |
|
‘Herbivores’ and primary predators |
Multicellular organisms as secon-dary predators |
In the Table, attempts are made to list events in chronological order, from the appearance of protobionts to that of secondary predators. Take note of the fact that some vacant niches/adaptive zones preexisted the emergence of organisms, while others were presumably created by organisms themselves (compiled from Lekevičius 2002).
As biomass accumulated, sooner or later aquatic resources of free organic compounds had to be depleted. That could have caused the rise of true autotrophs (photolithoautotrophs). The latter could have been green or purple sulfur bacteria, which used H2S and H2 as a source of hydrogen (electrons). Those bacteria accumulated sulfur and sulphates as waste, so after a while evolution should have brought about organisms reducing sulfur and sulphates. The vacant niche was occupied to make the cycle become non-waste again. After some time, however, the resources of H2S and H2 must have run short, that should have resulted in the appearance of cyanobacteria carrying out oxygenic photosynthesis. The merit of that kind of photosynthesis is in that it uses water molecules as a source of hydrogen (electrons). However, when oxygen turned into waste, it began to accumulate in water. As a result, the evolution of oxygen resistance was triggered off. Still after a while, presumably some 2.0–2.5 billion years ago, cyanobacteria and detritivores accompanying them became aerobes. It must have been at that time that all modern aerobic chemolitotrophs came into existence. The motives for their rise were very simple: oxygen accumulating in the environment reacted by itself with the dissolved in water ferrous iron and manganese, hydrogen, carbon monoxide, sulfur, hydrogen sulfide, ammonia, and methane. The energy produced during oxidation was lost. Naturally, those vacant niches became factors stimulating and directing evolution. Thus, after a while all those niches were occupied.
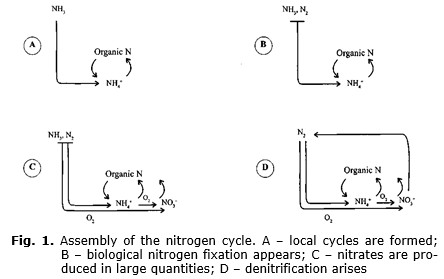
The nitrogen cycle was presumably
assembled in the following way (for details see in Lekevičius 2002). At the
dawn of life, nitrogen compounds, especially ammonia and ammonium ions, might
have apparently been much more abundant in the atmosphere and waters. Thus,
selection pressure, forcing organisms to acquire the ability of nitrogen
fixation, might have been absent for a while. Yet there are reasons to
believe that later the amount of ammonia and ammonium ions in the environment
decreased to minimum, and not only because part of it converted to organic
nitrogen, the biomass. Due to the presence of cyanobacteria, oxygen began to
accumulate in the environment and, affected probably by lightning, reacted with
ammonia and molecular nitrogen, thereby producing oxides. Besides, as it has
been mentioned above, soon thereafter originated nitrifying bacteria oxidizing
ammonia and ammonium ions to nitrates. I think that could have given rise to
selection pressure, which induced diversification of nitrogen fixing organisms
and their spread. Nitrates immediately created a vacant niche that provoked the
rise of denitrificators. The latter used nitrates as unchangeable under anoxic
conditions glucose oxidizers, final acceptors of electrons. Due to nitrate
respiration nitrates converted to free nitrogen. The global nitrogen cycle
became closed. Accumulating in the environment nitrates might have soon become
an additional source of nitrogen to cyanobacteria. Thus, we obtain the
following picture of the evolution of the nitrogen cycle (see Fig. 1). I
understand that this scenario of the changes in the nitrogen cycle is rather speculative, though it,
seemingly, is in accordance with the one proposed by experts (Falkowski 1997;
Raven and Yin 1998; Beaumont and Robert 1999). The difference lies merely in
some details of se-
condary importance.
There are sound reasons to believe that 2 billion years ago all modern global cycles – carbon, oxygen, nitrogen, sulfur – had been already formed. From the point of view of chemistry, they have not changed until nowadays (for details see Lekevičius 2002).
The stages of terrestrial ecosystem development and its mechanisms did not differ much from those of marine ecosystems (for details see Lekevičius 2002): appearance of producers (1), vegetative detritus (2), detritivores and local cycles (3), herbivores and organisms feeding on detritivores (4), primary predators (5), and so on up to the top-level predators. The latter came into existence in the late Carboniferous, approximately 300 million years ago. When the formation of the block of biophages finished in the seas and 135 million years later on land, there were almost no vacant niches left in ecosystems. Therefore cases of competitive exclusion, preconditioned by migration and the emergence of new forms, became more frequent. However, species diversification continued: life was penetrating into new territories, and what is more, the process of niche splitting was going on (Lekevičius 2002).
One may ask what there is new in such explanations of the well-known facts. In general, it is not customary in modern evolutionary biology to raise a question and to look for an explanation as to why certain guilds, let us say, aerobic chemolithoautotrophs or primary predators appeared on the evolutionary stage at that particular time and in that particular place. This can be probably explained by the fact that to find answers to questions of this kind, it is necessary to employ the deductive method, which is not popular with biologists. It has been only in this decade that somewhat wider, but still tentative use of the vacant niche term in the evolutionary theory has been started (Idem. 2009b). In case of failing to provide an explanation, a phenomenon itself is somewhat ignored. Another thing that makes my approach to evolution unconventional is that in respect of a population I emphasize external factors influencing the course of species evolution. Meanwhile, the conventional approach focuses all the attention on inner mechanisms. That does not mean, of course, that these approaches cannot be reconciled; they perfectly complement each other.
How Selection has Made Ecosystems Converge
The functional convergence of ecosystems was discovered quite long ago. Darlington (1957) wrote in his book Zoogeography: The Geographical Distribution of Animals:
Neither the world nor any main part of it has been overfull of animals in one epoch and empty in the next, and no great ecological roles have been long unfilled. There have always been (except perhaps for very short periods of time) herbivores and carnivores, large and small forms, and a variety of different minor adaptations, all in reasonable proportion to each other. Existing faunas show the same balance. Every continent has a fauna reasonably proportionate to its area and climate, and each main fauna has a reasonable proportion of herbivores, carnivores, etc. This cannot be due to chance (Darlington 1957).
Here I would like to draw the reader's attention to one important, in my opinion, episode from the history of general ecology. It is known that the ecosystem conception was developed on the basis of empirical data obtained in the 1960s of the last century. It was discovered, for instance, that neither the number of trophic levels, nor the ecosystem structure in general is dependent on primary productivity, which is known to vary within very great limits on a world scale. Fortunately for ecologists, nature turned out to be undivided, in that respect. Otherwise, it would have been necessary to develop individual conceptions for individual ecosystems. Thus, ecosystem convergence was a trivial fact for ecologists of that time.
Time passed and ecologists of the
older generation retired one after another to be replaced by young people
interested in other problems. That was possibly due to the fact that in those
times it was not easy to explain facts of the functional convergence of
ecosystems, since they were hardly within the framework of the neo-Darwinian
paradigm. It was difficult, or, according to somebody, impossible to build a
bridge between a change in gene frequency in a population and a global
phenomenon such as ecosystem convergence. It was ‘common knowledge’ that each
species is affected by a multitude of internal and external factors and that
its fate depends not only on an accidental genetic variability, but also on
gene drift, climatic changes that are usually difficult to forecast, the impact
of other species, and other difficult-to-define events. In the course
of millions of years, these numerous factors must have produced such chaos of
consequences in living nature that none of theorists was able to explain it.In a word, the opinion, which, by the way, persists to
date, was formed that evolution is controlled by accidental forces and that it
cannot be predicted. That is why the phenomenon of ecosystem convergence was
and is out of place in the neo-Darwinian conception. On the contrary, facts of
convergence contradicted the neo-Darwinian experience rather than supported it.
However, it is known that facts do not necessarily refute theories. It is often
the other way round – facts contradicting the generally accepted theory are
simply ignored. Thus, it is no wonder that in the course of time the interest
in that phenomenon gradually abated.
I propose using the notion of the functional convergence of ecosystems in a somewhat wider sense than that used by my colleagues some 20–30 years ago. I have in mind the invariability of ecosystem functions both in time and space. By this, I do not mean that ecosystems were not changing over time. I am inclined to take the view that approximately 2 billion years ago ecosystem metabolism finally became settled and since then nutrient cycles have been just replicated. The shape of production (energy) pyramids characteristic of local ecosystems seems to have undergone no considerable changes over the last several hundred million years despite all internal changes followed by numerous extinctions and adaptive radiations. Geographical invariance is also characteristic of these pyramids. Their form almost does not depend on the primary production, which may differ at least several ten-fold (the 10 % rule). Besides, when using the term ‘functional convergence of ecosystems’, it is necessary to have in mind the convergence at the level of individual species, too, i.e. a great abundance of ecological equivalents (species that have no consanguinity and live in different locations but have converged due to the fact that they occupy similar ecological niches).
As distinct from the traditional approach, I believe that all evolutionary processes are quite rigidly canalized. That role of canalization is performed by species interaction, which always and everywhere directs species evolution onto a few invariant ways. The raw material, from which evolution moulds a community, may differ. However, the final result, i.e. what the structure and function of that community is going to be like, is easier to predict because it often recurs both in time and space. God does not dice, so evolution could be predicted. But for this purpose, of course, one should have sufficient information not only about ancestral forms, but also about constraints. However, this information is as a rule lacking, because until today, in my opinion, evolutionists have not paid proper attention to factors constraining the evolution of species.
It is well-known that ecosystems may be assembled in two ways: via migration (ecological succession) and/or evolution. However, the final result does not depend on the mode of assembly, and that is evidenced by the fact of functional convergence. Probably, the same ecosystem-level constraints operate both in succession and evolution, although mechanisms are different. As a matter of fact, there are some similarities. Primary succession as a rule starts with the settlement of herbaceous plants (sometimes lichen). Then vegetative detritus is formed, niches suitable for the settlement of herbivores and detritivores (bacteria, protists, fungi and invertebrates) appear. As a result, necessary conditions for the appearance of soil are created (Olson 1958). In its own turn, the formation of soil stimulates the emergence of niches for new plants, woody plants among them. The latter change their surroundings, thus facilitating the settlement of still other plants and animals (facilitation theory – Connel and Slatyer 1977). The sequence of events is presumed to have been similar in the Palaeozoic when life occupied land (see above). However, then occupants came into existence mainly as a result of evolution in situ. So, I maintain that ecological succession may be interpreted as a process of niche filling as well, and it should not differ much in its course and final result (having in mind functional properties of ecosystems) from what is observed in cases of adaptive radiation and evolutionary recovery after extinction.
Odum (1969) put forward a hypothesis according to which ecological succession and evolution are characterized by the same trends of variation in ecosystem parameters (species diversity, primary production, total biomass, production and biomass ratio, efficiency of nutrient cycle). Although later this hypothesis was used as a target by many critically disposed opponents, it seems to be enjoying popularity among some ecologists and evolutionists (e.g., Loreau 1998; Solé et al. 2002; Lekevičius 2002, 2003) to date. In the opinion of these authors, forces directing the evolution of ecosystems are in fact the same as those controlling their routine action. Consequently, in both cases trends cannot differ much. This idea, that ‘ontogeny’ of ecosystems may recapitulate their ‘phylogeny’, I think, is quite attractive.
What is Selected vs. What is Making Selection
Extremist neo-Darwinians suggest that only the gene (‘selfish gene’) can be a unit of selection. Still others maintain that this role is more suitable for the genotype. Some evolutionists have claimed that differential survival may involve entire populations (species) and even ecosystems. Thus, there have been attempts not only to reveal mechanisms of individual features' evolution, but also to explain how parameters specific to populations and ecosystems could have evolved. So, there was hope to finally clarify how nature creates and maintains biodiversity and, on the basis of the latter, communities and nutrient cycles. Still others suggested combining all these ideas rejecting the mentality of ‘either-or’. Thus, the idea of hierarchic, or multilevel, selection emerged (e.g., Williams 1966; Gould 1982; Wilson 1997; Gould and Lloyd 1999). According to this idea, differential survival involves all or almost all structures ranging from single genes to entire ecosystems. As far as I understand, these evolutionists do not doubt that evolving are not only individual features, but also populations, ecosystems, and even the biosphere. However, they believe that adaptation at any level requires the process of natural selection operating at that level. I think that here they make an essential mistake for they restrict the problem of selection to the question of what is being selected. What is more, they seem to be little interested in what is making that selection. Because of that, the problem becomes quasi-complicated and, unfortunately, insoluble. I am inclined to think that Darwin, however, was right in assuming that it is an individual that should be regarded as the major unit of selection.
As far as I understand, the problem of selection units has become so complicated and intricate because it has not been associated with functional biology. The imaginary wall between biological time and biological space hinders researchers from finding a solution to this problem. If this wall was demolished, the problem would immediately become quite simple and clear. The greater the integration of constituent parts of a biosystem is, the greater the possibility is that selection will affect the whole system as a unit. And on the contrary, if constituent parts of a system are functionally autonomous, they will be involved into the ever-lasting ‘struggle for existence’ and each of them will become a selection unit. Even ecosystem selection would be possible, if ecosystems functioned as real superorganisms. However, this is inconceivable either for populations, or for ecosystems. By the way, the question of selection units was already solved in a similar way by Rosen (1967), but his point, apparently, has been overlooked.
In my opinion, natural selection is a ‘black box’ turning non-directional inheritable variability into a more-or-less directed evolutionary development (see Fig. 2). This is an essential attribute of selection. Differential survival and that kind of reproduction are merely external and most obvious features of selection. Quite possibly, selection may have another external form, too, but anyway it is the force constraining inheritable variability in a specific way.
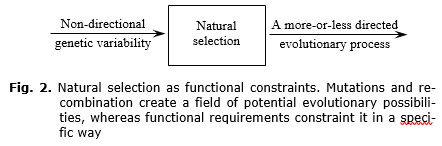
How does new genetic information in the form of a mutation or recombination become an attribute that changes the functioning of an individual, population and the entire ecosystem? Even the pathway of an especially successful mutation/recombination always begins from a single change in one of the cells. In the case of its success, novel genetic information passes several stages of strengthening. This may be done by means of the following mechanisms (Lekevičius 1986):
– transcription and translation of the newly emerged gene, increasing in concentration of mutant (recombinant) macromolecules in a cell;
– mitosis of cells carrying the gene;
– growth in the frequency of mutants (recombinants) in a population;
– growth of the population carrying the evolutionary novelty and widening of the species range.
Additionally, the variation has at least one more theoretical chance to be consolidated, which is to become the property of numerous species in the course of speciation.
As the novel genetic information is reinforced, an ecosystem reacts to it as to a gradually increasing internal disturbance. Individual – biochemical and physiological – mechanisms of adaptation are the first to respond. Mutants/recombinants are incorporated into adaptational and coadaptational processes at the population level after they pass barriers of internal selection. In case of success, new characters spread, but they have to prove they meet the requirements for constituent parts of an ecological community. If such co-adaptation happens, evolutionary diversification might follow and novel genetic information is disseminated among several or more species. In summary, the spread of evolutionary novelty always evokes feedbacks from individual, population and biocenotic mechanisms of adjustment, individual mechanisms being the first to react.
To sum up, traditional approach emphasizes selection units and cares about what is selected, whereas I propose taking interest in what is making selection. Differential survival and reproduction of individuals are merely external attributes and thus are the first impacts of adaptation to be noticed. It is functional constraints coordinating routine activities of individuals, populations and ecosystems that perform selection. They convert undirected hereditary variability into the far more directed evolutionary development. It is the individual that dies or produces fewer offspring, whereas structures, which may range from those of macromolecules to those of ecosystems, change and evolve. Moreover, competition is not necessary for the process of selection: it might be even more intense in the case of cooperation (e.g., features disturbing inner balance of an organism are done away via internal selection, or variations reducing the co-adaptation of sexual partners are also successfully eliminated). The difference is that in case of cooperation, only the characters beneficial to all cooperating partners are selected, while in case of competition, only the characters that enhance the adaptedness of particular competitors are selected. Of course, any novelty that is beneficial for the whole population or ecosystem, must be primarily beneficial for its possessor, only then it can be spread and, in this way, strengthened.
Concluding Remarks
During the past decade, strong nihilistic trends, far stronger than before, appeared in evolutionary biology. This is how one of the most authoritative evolutionists has summarized his approach:
Natural selection is a principle of local adaptation, not of general advance or progress. The history of life is not necessarily progressive; it is certainly not predictable. The earth's creatures have evolved through a series of contingent and fortuitous events (Gould 1994).
So, it turns out that Darwinism is suitable for the description of local phenomena of adaptation only. In this context it is worth remembering the previously published article by Gould and Lewontin (1979) expressing the authors' doubts regarding the whole adaptationist paradigm.
What way out do these authorities propose? Stephen J. Gould and Richard C. Lewontin seem to expect much from the theories of chaos, catastrophe, and complexity.
To describe that
situation I could find no better word than ‘crisis’. My opinion regarding the question is somewhat untraditional: biologists should reconcile
themselves to the idea that no one else will propose a suitable methodology for
the description of their subjects of study. A new methodology should take root
in the depth of biology itself. It should be sodden with biologists' sweat and
experience. None of the chaos, catastrophe, or complexity theories can or will
take root, like dozens of other exotic matters, for they have originated in
a different medium. If we do not want strange methods to dictate strange
to us objectives and world outlooks, we should assume the responsibility for
the future of biology. I disapprove of a further depreciation of mind and
reasoning, entrusting the function of thinking to the computer, being simply
afraid to form daring and audacious hypotheses that do not follow directly from
the data available. I dare to claim that the naked empiricism combined with
scientism raises monsters, i.e. young
people who, for the sake of solidarity, cut their own wings and burden
themselves with weights and lead in order to make their thinking as standard as
possible. I doubt whether Francis Bacon, the father of empiricism, would like
the scientific society so prone to standardize, but for me it is not very
appealing – it is my civic position if you like. I am for the balance of
induction and empiricism with deduction and rationalism rather than the
counterbalance between them as it is usually the case. I think that the method
of hypotheses advanced by Popper (1959) will be vindicated sooner or later. Bio-
logists should do this as soon as possible.
Acknowledgements
I am grateful to Michael Woodley and Laimutė Monkienė who helped me in translating and amending the text.
References
Beaumont V., and Robert F. 1999. Nitrogen Isotope Ratios of Kerogens in Precambrian Cherts: A Record of the Evolution of Atmosphere Chemistry? Precambrian Research 96: 63–82.
Connell J. H., and Slatyer R. O. 1977. Mechanisms of Succession in Natural Communities and their Role in Community Stability and Organization. American Naturalist 111: 1119–1144.
Conrad M. 1983. Adaptability: The Significance of Variability from Molecule to Ecosystem. New York – London: Plenum Press.
Darlington P. J. Jr. 1957. Zoogeography: The Geographical Distribution of Animals. New York: Wiley.
Darwin C. 1998 [1872]. The Origin of Species by Means of Natural Selection. 6th ed. Middlesex: Tiger Books Intern.
Falkowski P. G. 1997. Evolution of the Nitrogen Cycle and Its Influence on the Biological Sequestration of C02 in the Ocean. Nature 387(6630): 272–275.
Gould S. J. 1982. Darwinism and the Expansion of Evolutionary Theory. Science 216(4544): 380–387.
Gould S. J. 1994. The Evolution of Life on the Earth. Scientific American 271(4): 63–69.
Gould S. J., and Lewontin R. C. 1979. The Spandrels of San Marco and the Panglossian Paradigm: A Critique of the Adaptationist Programme. Proceedings of the Royal Society of London. Series B, Biological Sciences 205(1161): 581–598.
Gould S. J., and Lloyd E. A. 1999. Individuality and Adaptation across Levels of Selection: How shall We Name and Generalize the Unit of Darwinism? Proceedings of the National Academy of Sciences, USA 96(21): 11904–11909.
Koestler A. 1967. The Ghost in the Machine. New York: Random House.
Lekevičius E. 1985. Explanation in Biology: An Attempt to Synthesize Functional and Causal Approaches. Scientific Knowledge: Systematic Aspect / Ed. by R. Pavilionis, pp. 119–135. Vilnius: Institute of Philosophy, Sociology and Law. In Russian (Лекявичус Э. Объяснение в биологии: Попытка синтеза функционального и причинного подходов. Научное знание: системный аспект / Ред. Р. Павилионис, с. 119–135. Вильнюс: Институт философии, социологии и права).
Lekevičius E. 1986. Elements of the General Theory of Adaptation. Vilnius: ‘Mokslas’. In Russian (Лекявичус Э. Элементы общей теории адаптации. Вильнюс: «Мокслас»).
Lekevičius E. 1997. A Model of Adaptive Rearrangements on Community, Population and Organism Levels. Thesis for Habilitation. Vilnius: Institute of Ecology.
Lekevičius E. 2002. The Origin of Ecosystems by Means of Natural Selection. Vilnius: Institute of Ecology.
Lekevičius E. 2003. Evolution of Ecosystems: Main Stages and Potential Mechanisms. Zhurnal obshchei biologii 64(5): 371–388. In Russian (Лекявичус Э. Эволюция экосистем: основные этапы и возможные механизмы. Журнал общей биологии 64(5): 371–388).
Lekevičius E. 2009a. On Some Analogies between Ecosystems' Evolution and Economic
Development: From A. Smith and Ch. Darwin to the Newest Ideas. Evolution:
Cosmic, Biological, Social
/ Ed. by L. E. Grinin, A. V. Markov, and A. V. Ko-
rotayev, pp. 226–259. Moscow: LIBROKOM. In Russian (Лекявичюс Э.
О некоторых аналогиях между эволюцией экосистем и развитием экономики: от
А. Смита и Ч. Дарвина до новейших идей. Эволюция:
космическая, биологическая, социальная / Ред. Л. Е. Гринин, А. В. Марков,
А. В. Коротаев, с. 226–259. М.: ЛИБРОКОМ).
Lekevičius E. 2009b. Vacant Niches in Nature, Ecology, and Evolutionary Theory: A Mini-Review. Ekologija (Vilnius) 55(3–4): 165–174.
Loreau M. 1998. Ecosystem Development Explained by Competition within and between Material Cycles. Proceedings of the Royal Society of London. Series B, Biological Sciences 265: 33–38.
McLaren A. 1976. Mammalian Chimeras. Cambridge: Cambridge University Press.
Mesarović M. D., Macko D., and Takahara Y. 1970. Theory of Hierarchical Multilevel Systems. New York: Academic Press.
Novoseltsev V. N. 1978. Theory of Control and Biosystems. Analysis of Conservation Properties. Moscow: Nauka. In Russian (Новосельцев В. Н. Теория управления и биосистемы. Анализ сохранительных свойств. М.: Наука).
Odum E. P. 1969. The Strategy of Ecosystem Development. Science 164: 262–270.
Olson J. S. 1958. Rates of Succession and Soil Changes on Southern Lake Michigan Sand Dunes. Botanical Gazette 119: 125–170.
Popper K. 1959. The Logic of Scientific Discovery. London: Hutchinson.
Raven J. A., and Yin Z.-H. 1998. The Past, Present and Future of Nitrogenous Compounds in the Atmosphere, and their Interactions with Plants. New Phytologist 139: 205–219.
Rosen R. 1967. Optimality Principles in Biology. New York: Plenum Press.
Salthe S. N. 1985. Evolving Hierarchical Systems. New York: Columbia University Press.
Solé R. V., Montoya J. M., and Erwin D. H. 2002. Recovery after Mass Extinction: Evolutionary Assembly in Large-Scale Biosphere Dynamics. Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences 357: 697–707.
Williams G. C. 1966. Adaptation and Natural Selection: A Critique of Some Current Evolutionary Thought. Princeton, NJ: Princeton University Press.
Wilson D. S. 1997. Biological Communities as Functionally Organized Units. Ecology 78(7): 2018–2024.
